Neuherberg, 21.08.2020
A Gatekeeper Against Insulin Resistance in the Brain
The brain plays a major role in controlling our blood glucose levels. In type 2 diabetics this glucose metabolism brain control is often dysfunctional. Genetic components for this phenomenon have so far remained elusive. A group of scientists at Helmholtz Zentrum München and the German Center for Diabetes Research (DZD) have now shown in the ‘Journal of Clinical Investigations’ that in men a genetic variant of the gene DUSP8 can increase the risk for type 2 diabetes by impairing our brain response to the hormone insulin.
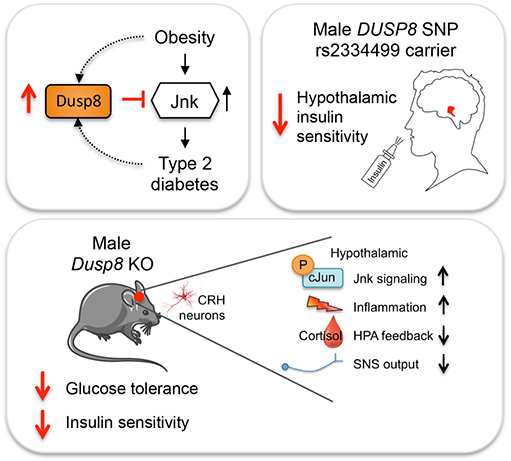
Insulin is a pancreatic hormone that controls our blood sugar levels. Insulin not only stimulates the uptake of glucose from blood into peripheral tissues, it also acts on the brain, specifically the hypothalamus, to control glucose and energy metabolism. In obese subjects, insulin loses some of its activity due to the activation of inflammatory signaling and a subsequent impairment of the cellular response downstream to the insulin receptor. This phenomenon, termed insulin resistance, is a major hallmark in the development of type 2 diabetes, but to date not yet fully understood. Recent genome-wide association studies (GWAS) identified DUSP8 as type 2 diabetes risk gene. Now, scientists have investigated how the protein Dusp8 (dual-specificity phosphatase 8), which is encoded by the DUSP8 gene, regulates glucose tolerance and insulin sensitivity.
Genetic variant of the DUSP8 gene increases risk of type 2 diabetes
“Carriers of a genetic variant of the gene DUSP8 were shown to have a moderately increased risk for type 2 diabetes, but the functional importance of Dusp8 for the etiology of the disease remained unknown,” explains Dr. Sonja C. Schriever, lead author of the study. “By combining cellular models, Dusp8 loss- and gain-of-function mice and functional magnetic resonance imaging (fMRI) of humans with genetic variants in the gene DUSP8, we now exposed a specific role of the protein Dusp8 as a gatekeeper for systemic glucose tolerance and hypothalamic insulin sensitivity,” adds colleague and senior author Prof. Dr. Paul Pfluger. Within the framework of the German Center for Diabetes Research (DZD), both scientists formed a team of biologists, epidemiologists and clinicians that was able to unravel the molecular mechanisms that link Dusp8 with the development of type 2 diabetes.
Protein Dusp8 has regulatory effects on hypothalamic insulin sensitivity
The protein Dusp8 has a regulatory effect on inflammatory processes in the hypothalamus and on the hypothalamic-pituitary-adrenal (HPA) axis. It seems to protect the organism against hyper-activation of inflammatory signaling and impaired insulin sensitivity in the hypothalamus. The scientists could show that the deletion of the Dusp8 gene in male but not in female mice increased hypothalamic inflammation, impaired HPA axis feedback and increased basal stress hormone levels, which all together aggravated the insulin sensitivity. The sex-specific role of murine Dusp8 was consistent with fMRI data in human volunteers that revealed reduced hypothalamic insulin sensitivity in male but not female carriers of the DUSP8 type 2 diabetes risk variant.
“Unraveling the multi-systemic processes that drive the impaired hypothalamic insulin sensitivity in the mouse models was an important step to understand the mechanistic underpinnings of the type 2 diabetes risk gene DUSP8”, concludes Schriever. In future studies, the researchers want to investigate the effect of central insulin action and the DUSP8 type 2 diabetes risk variant on the hypothalamic-pituitary-adrenal axis in human subjects with or without T2D.
Original publication:
Schriever SC et. al.: Type 2 diabetes risk gene Dusp8 regulates hypothalamic Jnk signaling and insulin sensitivity. J Clin Invest. 2020 Aug 11:136363. doi: 10.1172/JCI136363. PMID: 32780722.
Scientific contact:
Prof. Dr. Paul Pfluger
Helmholtz Zentrum München - German Research Center for Environmental Health
Research Unit NeuroBiology of Diabetes
and Neurobiology of Diabetes, TUM School of Medicine, Technical University of Munich
Ingolstädter Landstr. 1,
85764 Neuherberg
Phone +49 89 3187 2104
E-mail: paul.pfluger(at)helmholtz-muenchen.de
The Research Unit NeuroBiology of Diabetes (NBD) at Helmholtz Zentrum München studies the role of the central nervous system (CNS) as potential common denominator for obesity and diabetes type 2. NBD, which is associated with the IDO and part of the HDC and DZD, aims to understand the exact molecular underpinnings for leptin resistance and weight cycling and our adaptive physiological response to changes in the environment. www.helmholtz-muenchen.de/nbd
The Helmholtz Zentrum München, the German Research Center for Environmental Health, pursues the goal of developing personalized medical approaches for the prevention and therapy of major common diseases such as diabetes, allergies and lung diseases. To achieve this, it investigates the interaction of genetics, environmental factors and lifestyle. The Helmholtz Zentrum München is headquartered in Neuherberg in the north of Munich and has about 2,300 staff members. It is a member of the Helmholtz Association, a community of 18 scientific-technical and medical-biological research centers with a total of about 37,000 staff members. www.helmholtz-muenchen.de/en
The German Center for Diabetes Research (DZD) is one of six German centers of health research. It brings together experts in the field of diabetes research and combines basic research, epidemiology and clinical applications. Using a novel, integrative research approach, the DZD seeks to make a major contribution to developing a successful, personalized prevention, diagnosis and treatment of diabetes mellitus. The members of the network are Helmholtz Zentrum München – German Research Center for Environmental Health, the German Diabetes Center (DDZ) in Düsseldorf, the German Institute of Human Nutrition (DIfE) in Potsdam-Rehbrücke, the Institute for Diabetes Research and Metabolic Diseases of Helmholtz Zentrum München at the University of Tübingen and the Paul Langerhans Institute Dresden of Helmholtz Zentrum München at the TU Dresden Medical Center, associated partners at the universities in Heidelberg, Cologne, Leipzig, Lübeck and Munich as well as additional project partners. https://www.dzd-ev.de/en
Press contact

Birgit Niesing
niesing(at)dzd-ev.de
+49 (0)89 3187-3971